
Why is the ultra pure water resistivity of 18.2 MΩ•cm?
The conductivity is calculated as follows: X is the conductivity, F is the Faraday constant, Ci is the ion concentration, Zi is the charge of the ion, and Ui is the ion mobility. X = FΣ CiZiUi
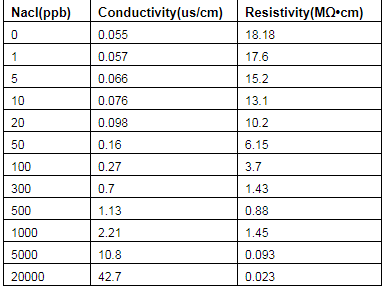
In theory, ultra-pure water completely free of ions has a conductivity of 0.055 μS/cm at 25°C. Resistivity is the inverse of conductivity. Then R=1/X, 1/0.055=18.1818, about 18.2 MΩ•cm
How does EDI remove organics?
EDI can only remove organic molecules that are charged.
Due to the voltage between the electrodes, micro-ionized organic molecules (such as acetic acid/oxalic acid/humic acid) migrate toward the anode.
For example: CH3COOH---CH3COO- + H+
CH3COO- is negatively charged and moves to the anode. The anion and cation membranes allow the anions and cations to accumulate in the concentrated channels. The water flowing through the electrode channels and the concentration channels of the EDI is abandonment of water.
Why is the terminal filter pore size only 0.22 microns?
The vast majority of bacteria are 1 micron in size. The smallest bacteria currently known are the pseudo-single strains grown under specific conditions. This bacterium is called Brevundimonas and its size is 0.3 micron.
Therefore, the use of a 0.22 micron end filter removes the smallest known bacterial strains.






